|
It is an intensive property, which is mathematically defined as mass divided by volume: Typical densities of various substances at atmospheric pressure.ĭensity is defined as the mass per unit volume. 
How does the atomic mass determine the density of materials? Density of Helium The atomic mass number determines especially the atomic mass of atoms. The mass number is different for each different isotope of a chemical element. For 63Cu, the atomic mass is less than 63, so this must be the dominant factor. A nucleus with greater binding energy has lower total energy, and therefore a lower mass according to Einstein’s mass-energy equivalence relation E = mc 2. The nuclear binding energy varies between nuclei.This increases the mass of nuclei with more neutrons than protons relative to the atomic mass unit scale based on 12C with equal numbers of protons and neutrons. 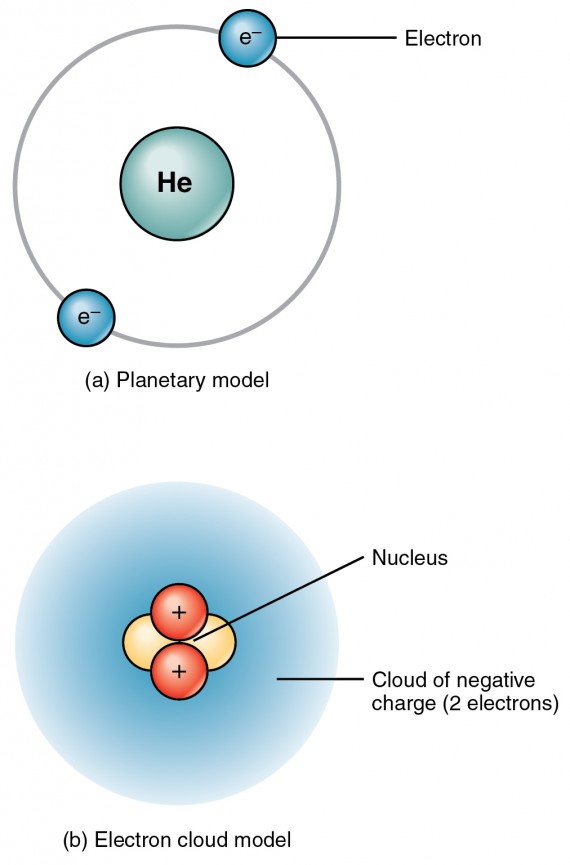
0 Comments
Leave a Reply. |

 RSS Feed
RSS Feed
